TOPCRYST
TOOLS FOR MACROMOLECULAR CRYSTALLISATION
FP7 – MARIE CURIE ACTIONS
INDUSTRY-ACADEMIA PARTNERSHIPS AND PATHWAYS
| The Method | The Partnership | Announcements | News |
|
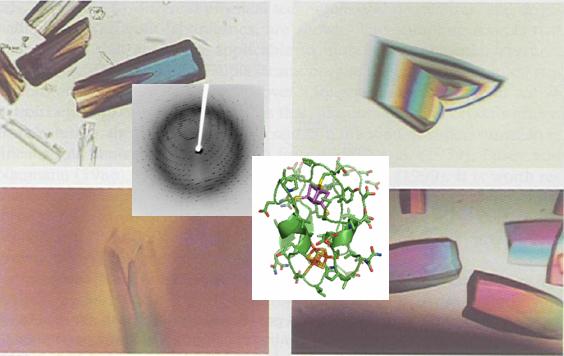
X-ray crystallography is the most widely used method for elucidating the three-dimensional structures of biological macromolecules, namely proteins and nucleic acids: the only method that allows macromolecular structures to be solved up to atomic resolution and without limitations on the size of the molecules studied. The knowledge of the atomic structure of such macromolecules and their complexes is of crucial importance for the elucidation of all the functions of living organisms, including the understanding of disease mechanisms at the molecular level. It is therefore of key importance for various applications such as:
Success in crystallographic structure elucidation depends absolutely on success in obtaining well-diffracting crystals of the molecule or complex. Crystallisation is the least controllable and usually rate-limiting step of the process that goes from cloning a gene to using the structural information for predicting and designing function. A lot of time and effort is spent on the quest for macromolecular crystals. In the majority of cases, these efforts fail. The great numbers of conditions that usually need to be blindly tried before promising results can be observed (if at all) and the corresponding requirements in large quantities of pure, highly concentrated macromolecular material, consumables, labour and time, are a major stumbling block to the process |
|